Background
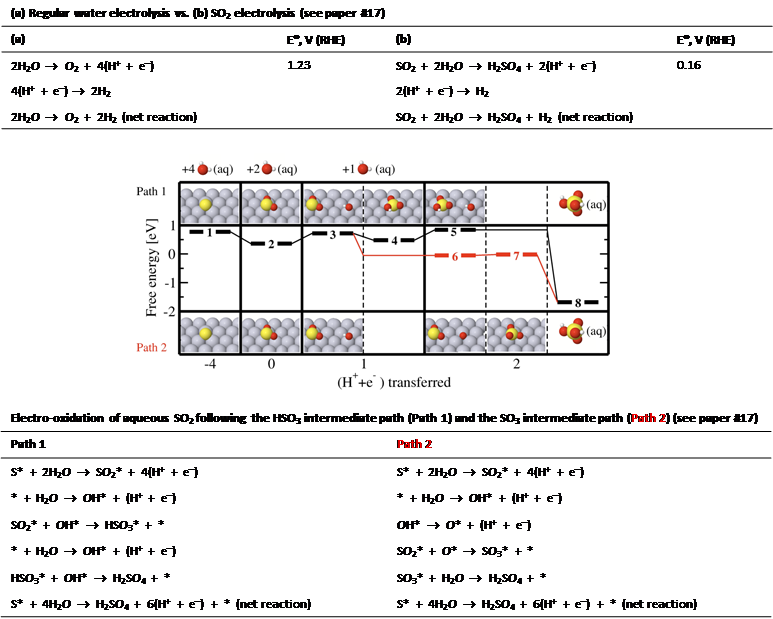
Our research focus includes investigating (i) vapour deposited thin metal films, nanoparticle metal oxide materials, and perovskite type materials for their electrocatalytic activity towards the oxygen evolution reaction (OER) in alkaline medium and the electro-oxidation of aqueous sulphur dioxide in acidic medium, (ii) membraneless alkaline water electrolysis, and (iii) the development of photo-electrocatalytic materials for water electrolysis.
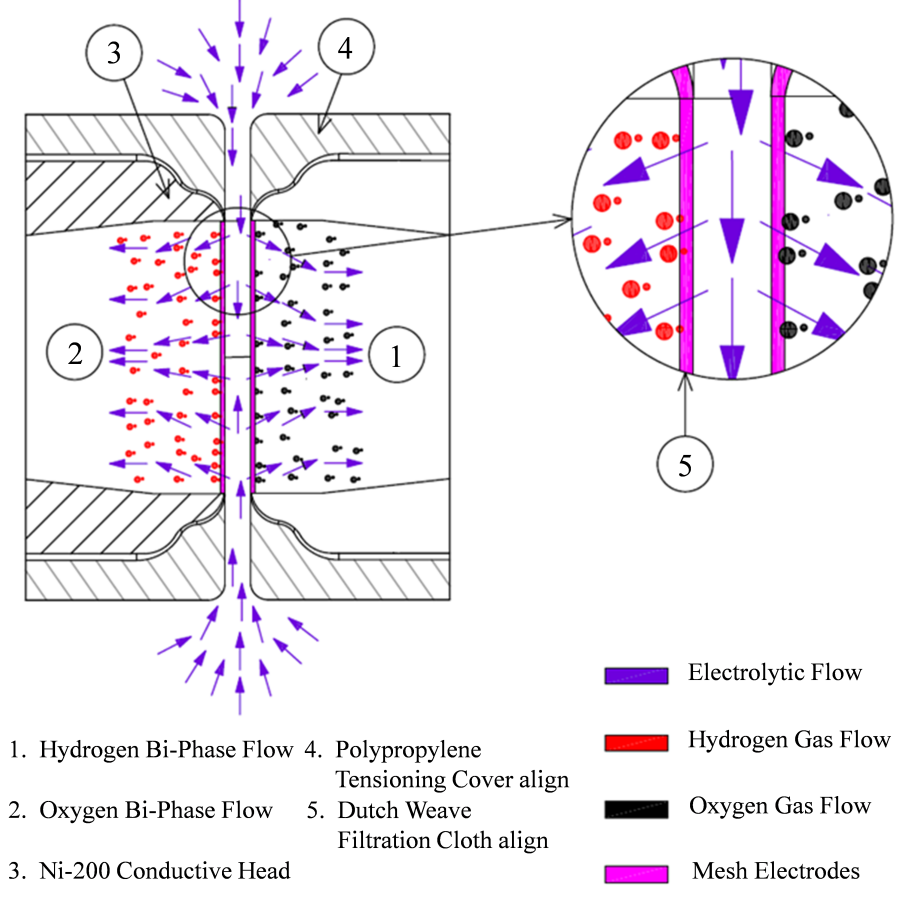
Divergent Electrode-Flow-Through (DEFTTM) tensioned circular mesh electrodes for membraneless alkaline water electrolysis (see paper #22)
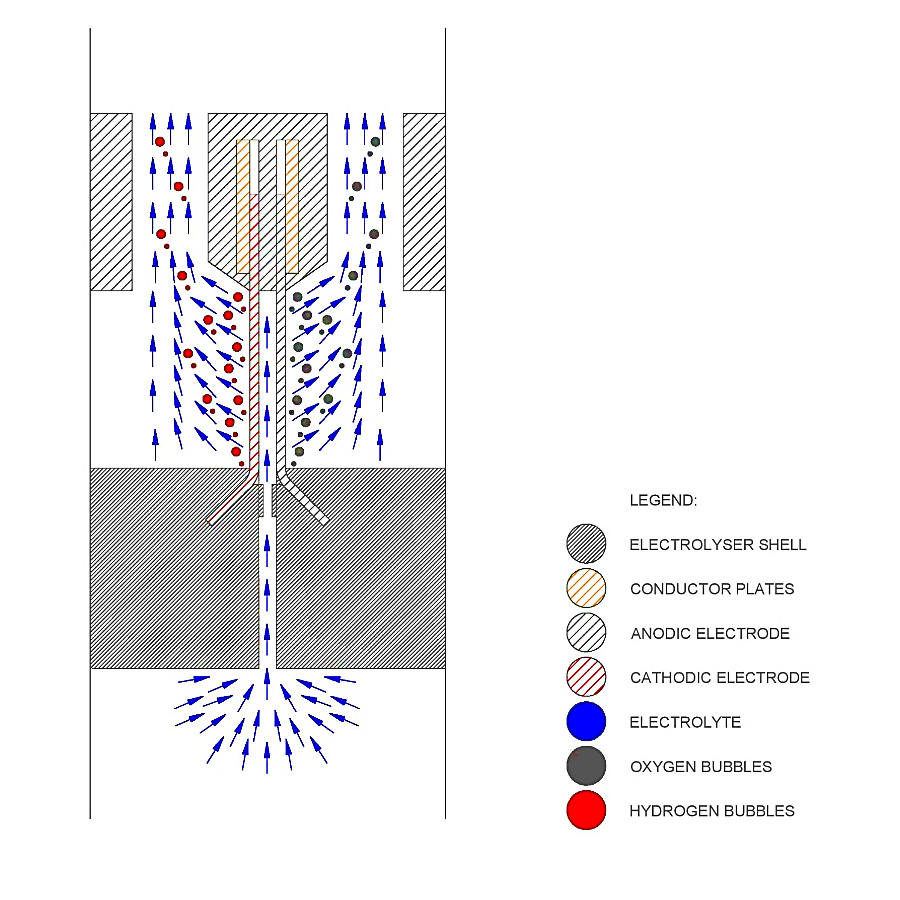
Divergent Electrode-Flow-Through (DEFTTM) tensioned rectangular mesh electrodes for membraneless alkaline water electrolysis (see paper #27)